Comparison and Selection of Exosome Analysis Techniques
Online InquiryExosomes, small extracellular vesicles derived from various cell types, have garnered immense interest in the field of biology and medicine due to their crucial role in intercellular communication and potential applications in diagnostics and therapeutics.
Isolation Methods for Exosomes
Ultracentrifugation
Ultracentrifugation, a time-honored method, involves subjecting biological samples to high-speed centrifugal forces, effectively separating exosomes based on their size and density. This technique comprises differential centrifugation and density gradient centrifugation.
- Differential Centrifugation. In this step-wise process, cell debris and larger vesicles are eliminated through successive rounds of centrifugation at increasing speeds. Exosomes, being smaller and less dense, sediment at lower speeds, yielding a highly enriched fraction. Creative Proteomics has optimized this method, minimizing contamination and maximizing exosome yield.
- Density Gradient Centrifugation. This advanced variant employs a density gradient medium, such as sucrose or iodixanol, to further purify exosomes. As the sample is layered on top of the gradient and centrifuged, exosomes migrate to their buoyant density position, allowing for precise isolation. Creative Proteomics' proprietary density gradient centrifugation protocols ensure the isolation of pristine exosome populations.
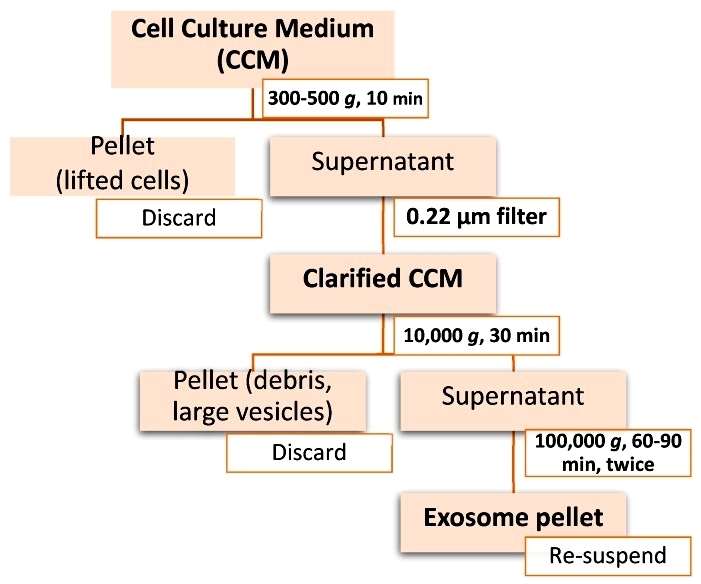 Workflow of differential ultracentrifugation for exosome isolation (Doyle LM, et al,. 2019)
Workflow of differential ultracentrifugation for exosome isolation (Doyle LM, et al,. 2019)
Size Exclusion Chromatography (SEC)
Size Exclusion Chromatography (SEC), a chromatographic technique, leverages porous columns to separate exosomes based on their size. As the sample passes through the column, smaller exosomes elute later, while larger contaminants exit earlier.
SEC offers distinct advantages, including rapid processing times, compatibility with various sample volumes, and minimal contamination risk. Creative Proteomics has harnessed the power of SEC to enhance exosome isolation efficiency, providing researchers with highly purified samples for downstream analysis.
Polymer-Based Precipitation
Creative Proteomics has also pioneered polymer-based precipitation techniques for exosome isolation. These methods capitalize on the unique physicochemical properties of polymers to selectively precipitate exosomes from complex biological fluids.
Polymer-based precipitation offers rapid processing, scalability, and high exosome yield. It obviates the need for specialized equipment, making it an attractive choice for large-scale isolation. Creative Proteomics' expertise in polymer-based precipitation ensures minimal interference from contaminants, resulting in pure exosome samples.
Choosing the Right Isolation Method
The selection of an exosome isolation method hinges on several factors, including the sample type, required purity, downstream applications, and available resources. Ultracentrifugation, with its versatility and robustness, remains a cornerstone in exosome isolation. For researchers seeking a balance between purity and speed, SEC presents an appealing option. Meanwhile, polymer-based precipitation caters to those pursuing rapid, scalable isolation.
Characterization Techniques
Transmission Electron Microscopy (TEM)
TEM allows for direct visualization of exosomes at nanometer resolution, providing insights into their morphology and size distribution. Creative Proteomics employs advanced TEM techniques to capture detailed images, aiding in the characterization of exosome samples.
Nanoparticle Tracking Analysis (NTA)
NTA quantifies the size and concentration of exosomes in solution by tracking their Brownian motion under a laser beam. This technique offers real-time analysis and is suitable for heterogeneous exosome populations. At Creative Proteomics, NTA is employed to precisely determine exosome size and concentration, contributing to accurate characterization.
Western Blotting
Western blotting helps identify specific exosomal proteins by targeting markers such as CD63, CD9, and Alix. Creative Proteomics employs Western blotting to validate exosome enrichment and composition, complementing other characterization methods.
Functional Analysis
RNA Profiling
Examining exosomal RNA content provides insights into their cellular origin and potential functions. Techniques like RNA sequencing and RT-qPCR enable the identification of miRNAs, mRNAs, and other RNA species. Creative Proteomics utilizes advanced RNA profiling methods to elucidate the role of exosomes in intercellular communication.
Proteomic Analysis
Mass spectrometry-based proteomic analysis enables the comprehensive identification of exosomal proteins, shedding light on their functional diversity. Creative Proteomics' expertise in proteomics allows for the identification of specific protein cargoes within exosomes, facilitating the understanding of their roles in various biological processes.
Selection of Techniques
Selecting the appropriate exosome analysis technique depends on the research goals, sample type, and desired outcomes. Creative Proteomics offers tailored solutions based on each client's specific needs. For comprehensive characterization, a combination of methods such as ultracentrifugation, TEM, NTA, and RNA profiling can provide a holistic view of exosome properties.
Learn more
In conclusion, the field of exosome analysis has witnessed remarkable advancements, with techniques ranging from isolation to functional characterization. Creative Proteomics stands at the forefront of these developments, offering cutting-edge services that empower researchers to unlock the mysteries of exosomes. By understanding the strengths and limitations of different analysis techniques, researchers can make informed decisions and pave the way for groundbreaking discoveries in biology and medicine.
Reference
- Doyle LM, Wang MZ. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells. 2019 ;8(7):727.
Related Services
* For Research Use Only. Not for use in diagnostic procedures.