Fortebio Octet Molecular Interaction Analysis Service
Online InquiryCreative Proteomics provides intermolecular interaction services based on the ForteBio Octet system, enabling quantitative and qualitative analysis of intermolecular affinity. The Octet RED system is a fully automatic test platform. Based on the biolayer interferometry (BLI) technology, only a small amount of sample is needed, and no labeling is required. It can provide real-time analysis results of biomolecular interactions. Compared with traditional GST pull down, co-immunoprecipitation, yeast two-hybrid, etc., it has the advantages of higher sensitivity, detection throughput and lower sample requirements.
The Princple of Fortebio Octet Molecular Interaction Analysis Service
The ForteBio Octet instrument has a biosensor inside. The bottom end of the biosensor is covered by a biofilm layer. The biofilm layer can bind and fix biomolecules. When visible light with a certain bandwidth enters the biofilm layer perpendicularly, the light forms an interference wave of a certain wavelength after being reflected at the two interfaces of the biofilm layer. When the fixed molecules interact with the molecules in the solution, the thickness of the biofilm layer increases, and the interference spectrum curve moves in the direction of increasing wavelength. The phase shift of the light wave is detected by the workstation in real time. Analysis of the phase shift of the light wave can quantitatively obtain the changes in the number of molecules on the sensor surface and the related concentration and kinetic data.

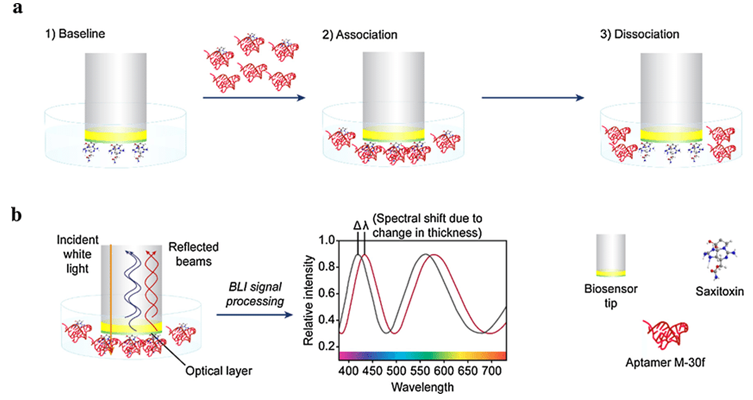 Schematic of The Working Principle of The Biosensor (Gao et al, 2017).
Schematic of The Working Principle of The Biosensor (Gao et al, 2017).
We Provide the Following Services, including but not Limited to:
- Qualitative analysis of intermolecular affinity: affinity report
- Quantitative analysis of affinity between molecules: binding constant (Ka), dissociation constant (Kd) and affinity constant (Kp); affinity ranking
Advantages of Fortebio Octet Molecular Interaction Analysis Service:
- High throughput: simultaneous detection and quantitative analysis of intermolecular interactions in the concentration range ng/mL to mg/mL.
- Wide range of applications: resistant to all kinds of solution environments, can directly detect crude samples, such as serum, plasma, tissue lysate, etc.
- Real-time measurement of kinetic data: the kinetic data of intermolecular interactions can be detected in real time. The transient interactions between molecules can be detected.
- Accurate results: precise quantitative analysis can obtain kinetic parameters to obtain more biological information. Non - marking, non - denaturation detection, more real response.
Sample Requirements
The detectable samples include: protein and protein, protein and small molecule, antigen and antibody, protein and antibody Fab fragment, DNA and DNA.
- The sample protein / antibody amount recommended is greater than 50ug.
- The buffer stored in the sample liquid does not contain Tris. PBS buffer is recommended.
Want to Know about Other Subcellular Protein-protein Interaction Analysis Techniques?
References
- Zuk R, Witte K, Heidecker B. Methods for characterizing molecular interactions: U.S. Patent Application 11/330,925. 2007-7-12.
- Nirschl M, Reuter F, Vörös J. Review of transducer principles for label-free biomolecular interaction analysis. Biosensors, 2011, 1(3): 70-92.
- Gao S, Zheng X, et al. Enzyme-linked, aptamer-based, competitive biolayer interferometry biosensor for palytoxin. Biosensors and Bioelectronics, 2017, 89: 952-958.
* For Research Use Only. Not for use in diagnostic procedures.