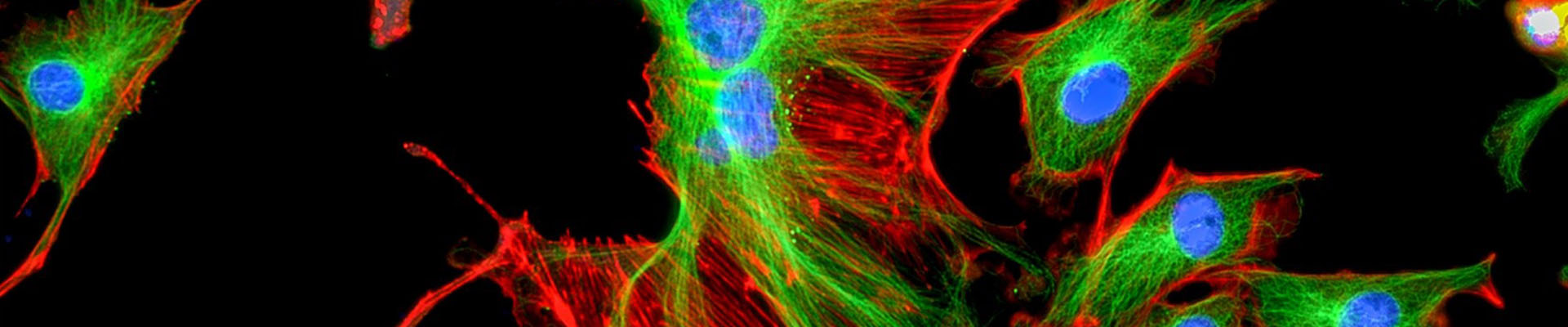
Fluorescent Protein Fusion Service
Online InquiryFluorescent proteins are ideal for live cell analysis because of their bright colors and intuitive images, which do not require chemical staining and substrates. Fluorescent tags that express proteins can not only label proteins in fixed and living cells, but also different cell types, and even subcellular organelles. If the target protein is fused with a fluorescent protein, intracellular observation of the target protein can be achieved, and it can be used for multicolor labeling and fluorescence resonance energy transfer (FRET). It is also used to visualize the location of proteins with other subcellular structures, study protein-protein colocalization, detect the onset of gene expression from different promoters, and isolate mixed cell populations. Creative Proteomics can provide fluorescent protein fusion services to accelerate your subject progress.
The Process of Fluorescent Protein Fusion Service
First, the gene encoding the protein in question is cloned and an expression vector is constructed. The gene sequence is fused with the coding sequence of a fluorescent protein (green fluorescent protein, GFP; yellow fluorescent protein, YFP; red fluorescent protein, RFP, etc.) to fuse the two genes into an open reading frame. Thereby, the protein of interest and the fluorescent protein are expressed in tandem. The obtained expression vector of the fusion gene is transformed into a target cell. Observe the localization of the fluorescent protein by fluorescence microscope or laser confocal microscope to determine the subcellular localization of the target protein. This method does not require specific antibodies for the protein of interest, but requires gene cloning and expression vector construction.
Problems You May Encounter During This Process
1. What color to choose?
The color is determined by the emission wavelength of the fluorophore. Color selection first requires selecting the correct absorption and emission wavelengths. If you want to do multiple experiments, you must make sure that the wavelengths of these several fluorophores do not overlap. The other is to think about your system. For in vitro experiments, most wavelengths are fine. In vivo studies, such as small animal imaging, only those long waves in the far infrared can effectively penetrate body tissues and play a role.
2. What do you want to mark?
The type of fluorescent label you choose also depends on what you want to label. That is, you have to consider the size of the fluorophore. For example, the dimerized Turbo series is more suitable for labeling cells and organelles and tracking promoter activity; the monomeric Tag series is relatively small and suitable for constructing fusion proteins for protein localization research and generating stable cell lines.
If you are still not sure how to choose, you can contact us. Creative Proteomics will advise you on your needs.
Advantages of Our Fluorescent Protein Fusion Service:
- We have fluorescent proteins covering a variety of spectral regions, including green fluorescent protein, blue and cyan fluorescent protein, yellow fluorescent protein, orange fluorescent protein, and red fluorescent protein. Different types of fluorescent proteins differ in brightness, light stability, and molecular size.
- One-stop service. The Creative Proteomics team will provide you with recommendations or test options based on your needs.
Application of Multiple Reaction Monitoring Protein Quantitative Technology
- Organelle research: GFP can be used to directly observe the number, distribution and movement of mitochondria in living cells during plant development.
- Protein research: Fusion the fluorescent probe with the protein of interest to study the distribution and changes of proteins in living cells.
Delivery
- Constructed vector map, complete sequence and annotation information.
- Sequencing results of the construction site.
- Sub-cell localization laser confocal finishing picture and original picture (optional).
- Experiment report (experimental steps and reagents, instruments and equipment used).
Want to Know about Other Subcellular Proteomics Quantitative Analysis Techniques?
References
- Zhang H, Linster E, et al. Tandem fluorescent protein timers for noninvasive relative protein lifetime measurement in plants. Plant physiology, 2019, 180(2): 718-731.
- Li C Y, Cao Y, Dewhirst M W. Applications of the Green Fluorescent. Handbook of Biomedical Fluorescence, 2019: 431.
- Von Arnim A G, et al. Cloning vectors for the expression of green fluorescent protein fusion proteins in transgenic plants. Gene, 1998, 221(1): 35-43.
* For Research Use Only. Not for use in diagnostic procedures.